2.5 The Third Law#
This law is an application of the second law. It states that the entropy of a perfect crystalline substance is zero at absolute zero (\(\pu{0 K}\)). The absolute entropy of a pure substance at a given temperature is the sum of all the entropy it would acquire on warming from absolute zero (where \(S=0\)) to the particular temperature.
Absolute entropy increases steadily with increasing temperature until the melting point is reached, where it jumps suddenly as the substance undergoes a phase change from a highly ordered solid to a disordered liquid (\(\Delta S_{fus}\)). The entropy again increases steadily with increasing temperature until the boiling point is reached, where it jumps suddenly as the liquid undergoes a phase change to a highly disordered gas (\(\Delta S_{vap}\)). As \(T\) increases, molecular motion increases, causing an increase in entropy (Fig. 33).
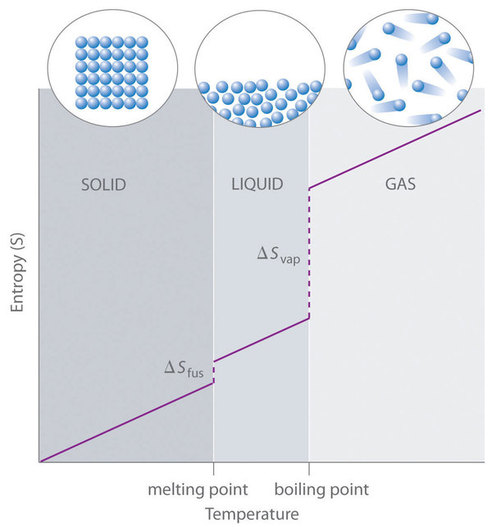
Fig. 33 A generalized plot of entropy versus temperature for a single substance. Image source: 13.6: The Third Law of Thermodynamics | LibreTexts#
The third law of thermodynamics has two important consequences: it defines the sign of the entropy of any substance at temperatures above absolute zero as positive, and it provides a fixed reference point that allows us to measure the absolute entropy of any substance at any temperature.
All entropy values of substances are absolute entropies at a given \(T\) compared with their corresponding values (\(=0\)) at absolute zero. Absolute entropies are standard entropies (\(S^\circ\)) at \(\pu{1 atm}\).
