7.3 Practice Problems#
Assume that in 1951, before the first atmospheric tests of fusion bombs, the concentration of \(\ce{^3H}\) in rainwater was \(\pu{8 TU}\). Calculate the concentration for pre-bomb in a present-day groundwater reservoir. \((\lambda = \pu{5.575e-2 y-1})\) Note: \(\pu{1 TU} = 1\, \ce{^3H}\) per \(\pu{e18}\, \ce{H}\) atoms.
\(\ce{^{14}C}\) activity of modern-day \(\ce{C}\) is \(\pu{13.56 dpm g−1}\). If charcoal from an ancient fire has an activity of \(\pu{6.22 dpm g−1}\), how old was the fire? \((\lambda = \pu{1.209e-4 y-1})\). Note: dpm refers to disintegrations (of the radioisotope) per minute.
\(\ce{^{137}Cs}\) is often used to radio-date fresh surface sediment, and it’s \(t_{1/2} = \pu{30 y}\). From an original sample of \(\pu{80 g}\) of \(\ce{^{137}Cs}\), how much is left in \(\pu{80 y}\)?
\(\ce{^{230}Th}\) activity in surface of a marine sediment core is \(\pu{62 dpm}\) and \(\pu{28 dpm}\) at \(\pu{1 m}\) depth. \((\lambda = \pu{9.217e-6 y-1})\).
What is the age of sediment at \(\pu{1 m}\) depth?
What is the sedimentation age in \(\pu{m y-1}\).
A water sample has a \(\delta \ce{^{18}O}\) value of \(\pu{−4.5 ‰}\). It is in equilibrium with water vapor at \(\pu{25 ^\circ C}\). The fractionation factor at \(\pu{25 ^\circ C}\) is \(1.0093\). What is the \(\delta \ce{^{18}O}\) value of the water vapor? %Answer: \(\pu{−13.67 ‰}\)
For Greenland, average summer temperatures are \(\pu{0 ^\circ C}\) and average winter temperatures are \(\pu{-30 ^\circ C}\). The fractionation factors for \(\delta \ce{^{18}O}\) are: at \(\pu{-30 ^\circ C}\), \(\alpha = 1.0155\) and at \(\pu{0 ^\circ C}\), \(\alpha = 1.0112\). Calculate the isotopic composition of snow formed during the summer and winter seasons. %Answer: Summer = \(\pu{−24.4 ‰}\) and winter = \(\pu{−29.9 ‰}\)
A water sample was measured by isotope ratio mass spectrometry to have a \(\ce{^{18}O/^{16}O}\) ratio \(R = 0.00199517\) with VSMOW (\(R = 0.00200520\)). What is its \(\delta\) value (rounded to one decimal place)?
Identify which water sample is isotopically lightest in the figure below. Provide an appropriate rationale.
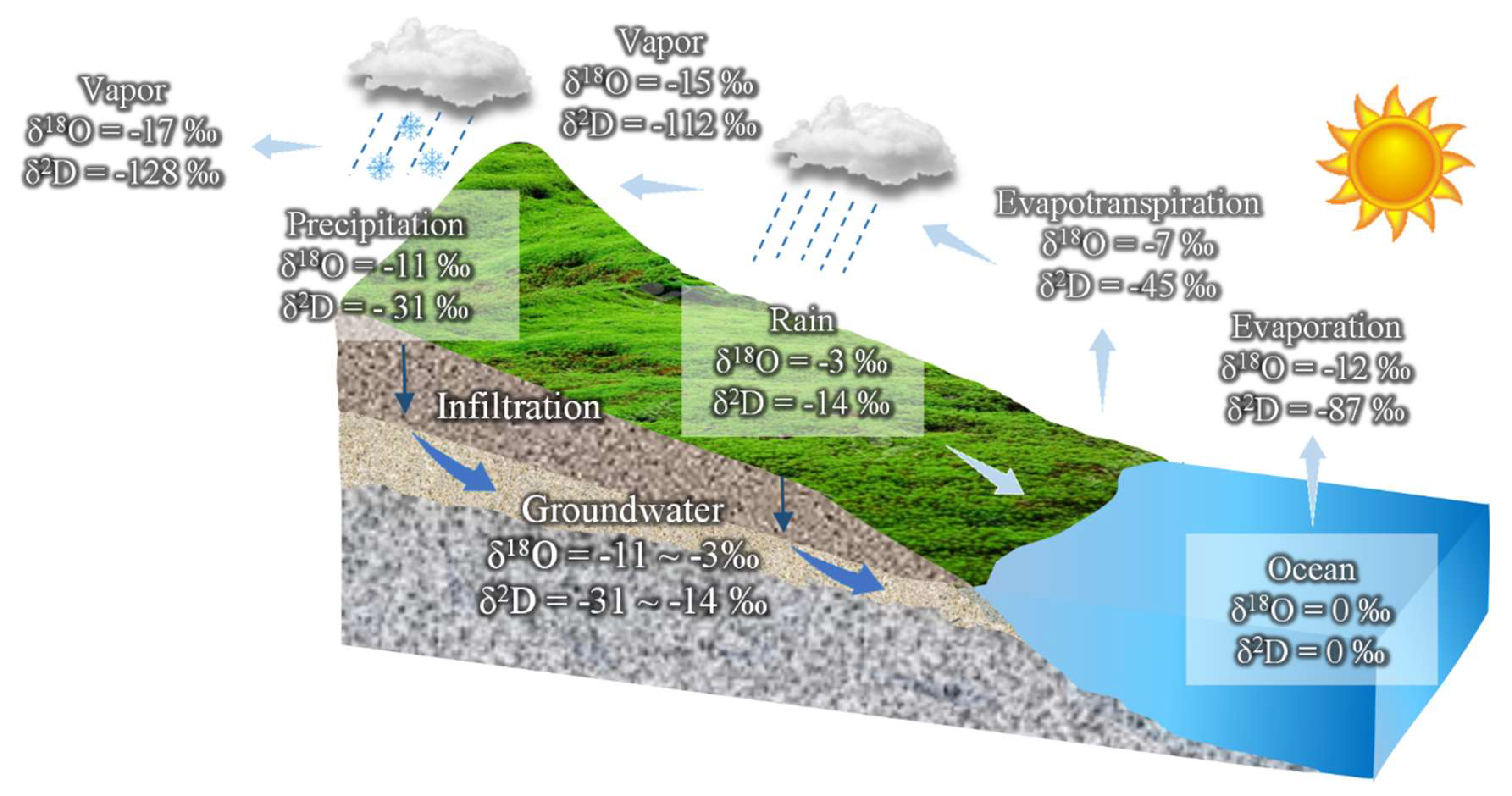
Fig. 56 The diagram of the isotopic composition change of atmospheric water vapor shows the processes of evaporation and rainout as the air mass proceeds from an ocean to a continent. Image source: Stable Isotopes of Water and Nitrate for the Identification of Groundwater Flowpaths: A Review (mdpi.com)#
